 This week featured me wrapping up all of the known aspects of this final chapter of my dissertation. What I mean is that I finished counting all the meiofauna and I will finish their biomasses this weekend. After nearly 4000 individual animals, the meiofauna or known part is done. The next step is the unknown--the macrofauna. Macrofauna are the organisms that exist one size class above meiofauna. Where meiofauna are commonly defined as organisms with body sizes 32-500 micrometers, macrofauna often exist between 500 micrometers and 5 cm. While these size ranges shift depending on the environment (deep sea shifts toward smaller organisms), the rough size classification system is based on sieve mesh sizes, which is why the larger mesh size that I use in my research is 500 micrometers. This larger size retains all organisms at 500 micrometers and larger, while the meiofauna pass through the mesh and into the smaller sieve below. Once we separate the macrofauna and meiofauna by sieve size, we transfer the macrofauna to sampling jars, add a fixative, usually formaldehyde (formalin), and then store the samples away, often to never see the light of day again unless a researcher is interested in those organisms. Therefore, we do not have macrofauna expertise nor guidebooks in our lab for identifying these animals. While we can likely identify the animals to very broad taxonomic classifications (think identifying a monarch butterfly as an insect and no further), the data I need for my experimental analysis has to be more rigorous (think identifying a monarch butterfly as Genus: Danaus or as Danaus plexippus). The specificity in this chapter of my dissertation is important, as I will assign each animal a mobility and a sediment reworking score based on its life history traits. These scores have been defined by other scientists so the closer I can get to their level of specificity, the better the accuracy of my classifications. Next week I will be taking a week off from the blog as I use next week to take a short mental health break. This work has been quite tiring and I want to ensure that my work is reflective of the scientist I am, rather than reflective of my level of focus. I will spend some time identifying some of the remaining unknown meiofauna from the remaining organisms, including this little critter that looks like a fun microscopic fire hydrant. See you in two weeks.
0 Comments
 This week in the lab I have been making microscope slides and taking more pictures to complete the biomass measurements for the meiofauna. I've resorted to calling the biomass process 'clicky clacky' as I have to click the mouse so many times to complete one measurement I'm afraid that my office mates are losing their patience. Why so many clicks? The ImageJ program has multiple methods for measuring photographs. The two most common methods are using a straight line tool and a segmented line tool, which as its name suggests, is used to measure items not in a single straight line. Since many nematodes exist in non-linear shapes, and nearly all copepods have segmented shapes, I have to click multiple times to connect line segments from their heads to tails. Additionally, ImageJ keeps track of all measurements I take in a small side window, however it assumes that each measurement is from a separate animal, so all the measurements regardless of length or width, are stored in one column. Therefore, when I import the measurements into Excel, I need to extract the width measurements, make them their own column, and then align them with the associated length measurements; again, more clicking. I am pleased to note, however, that today I finished slides for sample 24 out of 32. Between today and Tuesday--my next day at the lab--I will complete 12 samples of biomass measurements to catch up to my slide making. This means that next week will likely be my last week of slides and meiofauna biomass. I still have a few animals that I have to identify, as they are taxa I'm not familiar with, and a few copepods I have to look at under the microscope and compare their shapes with reference material (there are 8 copepod shapes which each require different conversion factors). Once this process is done, I'm one step closer to finishing this research. My interim step will be converting all of the biovolume measurements to biomass measurements using the formulas and conversions I talked about in last week's blog. Then I can move on to the macrofauna, which will be an easier biomass process, though a more difficult identification process, since we don't work with macrofauna in our lab and we don't identify them. Maybe Google reverse image search will have some ideas. This week at the lab I moved onto the next phase of wrapping up the mesocosm experiment: biomass measurements. Biomass, as the name suggests, is the weight of any organism, and this metric is common in fields of ecology and agricultural science. While most scientists collect biomass measurements by weighing the organism and then drying the organism out to get a dry weight, this process is often not suitable for meiofauna research given the size of organisms involved. A quick Google search tells me that larger individual nematodes can weigh between 0.04 and 8 micrograms, which would be painstaking to mass each individual. Instead, researchers developed a suitable biomass calculation for all meiofauna individuals. This method involves using biovolume measurements and conversion factors to calculate biomass. Here's how the process works: 1) individual animals are either photographed or mounted on slides for microscopic analysis; 2) body measurements are collected from the animal, usually length and width measurements, from the photographs or from software connected to the microscope; 3) biovolume measurements are calculated by multiplying the length x width^2 x conversion factor, and these conversion factors are specific to the types of animals and vary based on body shapes for some animals; 4) finally, these biovolume measurements are converted to wet weights and then dry weights using additional conversion factors, which have been validated by researchers.
At the lab this week, I finished extracting all the meiofauna from my samples and I am currently making slides for all the animals. During the slide making process, I am also photographing all the slides to take biovolume measurements. This photography and calculation process is made much easier since the microscope at the lab has a fixed zoom, where I can set the zoom and it will not move, guaranteeing consistent distances to compare to the reference photograph for scale. The photographic measurements are also quite helpful since I can upload the photos and complete the measurements elsewhere, rather than staying at the lab extra hours during the week. The entire process of making slides, photographing samples, and collecting biomass measurements for the meiofauna will likely take the entirety of October, although using a photographic analysis rather than the in-person microscope software analysis saves a few weeks of time. Once I finish the meiofauna measurements I will move on to the macrofauna, which will be a whole other beast and a different biomass measurement process altogether. Stay tuned! This week was a slow week thanks to the brain break that I took, and probably really needed. I couldn't be at the lab one of my usual two days due to other scheduled events, and today I didn't feel as full of energy as usual. Therefore, rather than pushing through and possibly missing fauna during the counting process, I took a step back, did fewer samples, and took a break. I will get back to the samples on Tuesday, after this much needed mini break. While this break means I'm slightly behind, I'm not worried, as I only have two more samples to pick and count and then I will be moving on to biomass measurements--the focus of next week's blog.
October and November, possibly December too, will be dedicated to biomass measurements, and I'm hoping to have approximate a timeline for this work soon. Additionally, October means slightly cooler weather, as we have been dealing with temperatures in the high 80s and 90s for too long, and I am hoping for true Fall weather rather than the horrid temperatures that plague the American southeast. I'm grateful for no snow, but wishing for either days of rain or at least cooler air so that I can enjoy the Fall. Tune in next week for an explanation of the biomass process. For those of you who are familiar with measuring the biomass of larger animals or plants, you may be surprised how we conduct this work on microscopic animals. See you then!  This week's blog is dedicated to the true heroes of this chapter of my dissertation research. They are small and mighty, but they have really pulled their own to make this chapter run as smoothly as it has. The first, pictured here, are the 1% of all the animals I've picked and counted that have retained the Bengal Rose stain. You may remember from a few weeks ago that the animals did not retain the stain initially, so I tried increasing the stain concentration. Well, to no avail, as only approximately 1% of all the animals have retained even this concentrated version. Seeing one or even two stained meiofauna in a sample is a sight for sore eyes, as they are easier to track in the picking and transferring process than their translucent counterparts. The other hero of the week, or really of the entirety of my lab work, is my music. I know that as a student I heard that working in a lab meant accepting your supervisor's or PI's music and I was told stories of listening to endless hours of the radio. Luckily for me, our lab doesn't have communal music players or speakers, so I can sit down, put my headphones in, and listen to my podcasts (of which there are so many), music, or audiobooks. I know that Dr. Ingels gets a chuckle when he walks by the lab and I'm bopping my head to music and sometimes I am laughing at the things my podcasts talk about. Most of the time, my podcasts and audiobooks aren't academic, so I don't feel like I'm adding extra work to my day, and I can sit, focus on my task, and have enjoyable media playing in the background. I've even had to put up a sign on the lab door explaining that if I have my headphones in, I likely can't hear people at the door so they need to come tap me on the shoulder to get my attention. So here's to the true, unsung heroes, who have gotten me through these past few weeks. As of today, I have sorted through 26 of my 32 mesocosm containers, and picked out all their meiofauna. I'm hoping to finish the final 6 next week and move on to measuring all these animals. Expect a big blog explaining the measuring process soon! 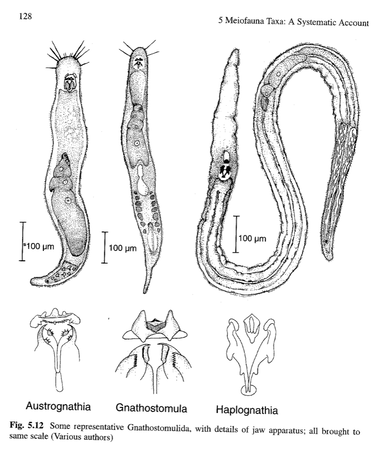 This week has been all about picking out more animals in the mesocosm muds, and I am excited to share a bit about three new animals I have discovered. When I say, 'discovered' here I don't mean that I am the first scientist to observe these animals. Instead, I mean that these animals are quite rare in my work, so I am excited to find them. When I started my dissertation and my time at FSU, I focused largely on two meiobenthic taxa: nematodes and copepods. These taxa are quite abundant in marine sediments, so I was very much aware of them throughout all of my samples. Whether I was looking for microplastics, or picking out animals, I recognized the nematodes and copepods. For the second portion of my dissertation, I learned to identify nematodes to the genus level using taxonomic keys and I learned how differences in nematode communities are representative of environmental health and disturbances in the sediment. In my microcosm experiment, I analyzed every animal I found to see if it had eaten microplastics during the experiment, but these animals were mostly nematodes. Of the nearly 10,000 animals I assessed during this work, nearly 99% were nematodes with a few copepods, kinorhynchs, polychaetes, isopods, nemerteans, and rotifers. However, this week, I found three new (or newer) taxa, that I have not had experience working with. One of the three I have seen before, but its name is escaping me, and a quick look through the visual guide didn't lead me to any conclusions. The second taxa is brand new to me, and it looked unlike any fauna I have seen or read about, so I will have to hunt down some resources to help identify the animal. The third taxa, shown here, I believe is gnathifera, which are quite fun little animals. They remind me of a drawing a child would make if you asked them to combine a potato and a worm and then make it very small. Even the taxonomic guide calls them "more or less cylindrical," which is quite apt. Gnathifera, specifically gnathostomulids, are associated with fine sands that are detritus (dead matter) rich, hypoxic (very low oxygen), or sulfur-rich. While I'm not sure which of these features best describes the mud they came from, they are a real treat and add some novelty to my day of sifting through hundreds of nematodes. This week I finished washing all 32 of the mesocosms and separated the macrofauna and meiofauna from the residual sediment. Therefore, I moved on to step two in the sediment processing, which is the counting and analysis phase. While I only looked through five samples this week, I am happy to learn that the samples are not packed to the brim with fauna. Going through each meiofauna sample took between 1.5 and 2 hours, with each sample having similar numbers of animals. It will take me longer to process samples with more animals, of course, but this current pace is acceptable in my opinion. The one issue I've noticed is that none of the fauna were stained even though I added the stain to all of the samples. This means that every sample I looked at today had translucent fauna, which increases the amount of time it takes me to process the samples, since the animals are translucent rather than pink/red. I added an extra drop of stain to all the samples before I left today, so I'm hoping to get back to the lab next week and have colorful samples. Otherwise, I'm going to be stuck with a difficult few weeks ahead of me.
Step two also involves slide making, so I will spend every other day at the lab mounting the animals on slides. Rather than processing and picking out animals from every sample and then making slides all at once, I go back and forth between these two processes to limit the amount of lab materials I am using as we have a limited amount of containers and space to store all these materials. Additionally, going between counting and slide making helps me make the most use of my time, as I can usually process an extra sample during the week by going between these two steps. My goal is to have the meiofauna counted and slides made for all samples by mid-October. After I make all the slides, I will likely move to making slides for the macrofauna samples, which should go faster since the organisms are larger and won't require the same precision searching as the meiofauna samples. Sadly, no picture this week, as I couldn't find the microscope's SD card and by the time I realized it wasn't in the microscope, I had finished going through my last sample for the day. Next week's blog will definitely come with a photo, maybe of an unexpected animal in a sample, because who knows what I will find. Stay tuned!  After a one-week research hiatus, thanks to Hurricane Idalia (which didn't cause damage to my area in Florida but threatened to), I was back at the lab this week working on the next phase of the mesocosm research. Like the first chapters of my dissertation, I have to wash all the samples and extract all the fauna, though it's more time consuming now given that I have more sediment than usual (7 versus 5 cm in each sample). Additionally, a new aspect of this work is an emphasis on the whole sediment community rather than a focus on the meiofauna size class. Macrofauna, animals slightly larger than meiofauna--usually 500 micrometers and larger (up to a few centimeters)--perform many of the same bioturbation activities as their smaller counterparts. Therefore, their presence in the sediment communities and specifically the number and size of these animals can drastically shift the bioturbation activity across the sediment communities. Since I used natural sediment communities for this experiment, communities collected from individual sediment core deployments, I don't know that the starting communities were all the same and the differences within these communities may have contributed to differences in the bioturbation and oxygen penetration within the experiment. Rather than an extra byproduct of my research, these fauna are an integral component of the data analysis for this final chapter of my dissertation, and you can see the start of the sample processing in this week's photo. I will complete sediment washing and processing next week, after which I move on to the next step which is counting, slide making, identifying, and measuring. This next step will help set the timeline for the completion of this work, as the number of fauna within each sediment sample and their identities will inform how long biomass measurements will take to complete. Additionally, next week will mark the end of what I consider the most exhausting part of my work which is sediment washing. Standing for hours in the lab using the spray nozzle to gently wash the mud and make sure the water runs clear is often a taxing process, and even though I don't love sitting at the microscope, I do find the work more exciting and less monotonous, possibly because going through samples becomes a game of 'I Spy.' I am grateful that for this chapter I am again able to stain the meiofauna samples with the Rose Bengal stain (the pink colorant you've seen in past photos), as the stain is extremely helpful in visualizing all the animals within the sample. Never again do I want to design a project where staining the individuals is not an option. Tune in next week as we explore the first set of fauna samples from the final chapter of my dissertation!  After two weeks of data collection and working desperately to maintain the environmental conditions we set in the experimental chamber, Adele and I have finished our mesocosm experiment. This experiment had some unexpected challenges at the end, largely because the ocean conditions during sediment collection were so warm, that we had to maintain those same conditions in the environmental chamber. These warm conditions meant that the air conditioning system couldn't effectively dehumidify the chamber, so our microsensors were challenged right until the end. While the data collection aspects are complete, I still have work to accomplish before I can claim this experiment is fully finished. Since each mesocosm came from an individual sediment core in the field, each mesocosm represents a community of organisms that may not be the same as the rest of the mesocosms. Therefore, I need to identify and count every single animal present in the mesocosms in case differences in the animal communities caused differences in either the oxygen penetration or the bioturbation. This process, along with collecting their biomass measurements, will take a few months and I am hoping to wrap up this phase by winter break in December. In the meantime, I will be writing up some initial results to send to the Ocean Sciences conference committee in hopes of presenting this work at the Feb 2024 Ocean Sciences meeting in New Orleans. This conference is the flagship conference for the ocean sciences and is a great opportunity to share interesting work and to network with professionals and meet other students. By the time the conference happens, I will be done analyzing all the data for this work, so it will make for a great time to talk about this cool research I did and how microplastics may alter benthic ecosystem functions. Not the most beautiful picture to close out the blog this week, but instead, a ducks-in-a-row picture of the former mesocosm containers (left), macrofauna containers (middle), and meiofauna tubes (right) all ready for sample processing which will start next week.  After a few minor hiccups regarding the mesocosm setup, Adele and I are off to a great start with this research. Adele, a PhD Candidate at University of Goteborg in Sweden, arrived last week after an exhausting day of travel to help me with this final project. As an expert in marine biogeochemistry, particularly in marine sediments, Adele is well-suited to collect the sediment oxygen data for this work. The minor hiccups actually regarded some of the setup for her materials, as Adele traveled with nearly $10,000 of research equipment and one piece wouldn't fit in her luggage so she asked me to build it (thanks dad for the great DIY version). This DIY piece of equipment shown in this week's picture, is meant to hold the micromanipulator so that the oxygen microelectrodes--tiny needle-like sensors that measure oxygen profiles in the mud--can enter the samples vertically. The micromanipulator is then programmed to slowly lower the microelectrodes into the water or sediment (.001 millimeters at a time in some cases) and take measurements at set intervals that are logged by the computer not shown in this picture. By collecting these oxygen measurements, we learn how oxygen availability changes in sediments when microplastics are present and we can also calculate the oxygen penetration depth, the maximum depth that oxygen is available within the sediment. In this picture you can also see the luminophores in action, though they look like radioactive material because of their bright green color. On the other side of the environmental chamber, I am taking pictures of each sediment mesocosm after exposing the sediments to UV light and tracking how deep the luminophores travel. These two aspects of the experiment--oxygen penetration and bioturbation--are inherently linked processes and important ecosystem functions provided by the animals that live on or in the sediment. Our goal is to learn how microplastics alter these processes using concentrations of pollution from the field and expected future concentrations. It has been such a joy working on this project, problem-solving through the bumps in the road, even sitting at the computer to process all these images. Adele and I will be collecting data for one more week and then she will return to Sweden while I finish the last data collection components--more on that in a future blog. Stay tuned, because next week will mark the end of the experiment so you know I will be writing an exciting blog to wrap up this portion of the work. Not a big update for this week, but Adele, my collaborator from Sweden who is an expert in marine biogeochemistry, arrives later this evening so that we can start this final project either tomorrow or Saturday, depending on how we are feeling and if everything is working as it should. Once the experiment starts, we will let it run for about two weeks and we will collect data every other day to track bioturbation and oxygen turnover in the sediments incubating at the lab. As this project is pretty intensive with regards to the time commitment, I will be staying over at the marine lab, which will provide quick access to the experimental set up and saves me a 2.5 hour drive daily (that's time spent driving there and back combined, not separate). Over the next two weeks we will hopefully have an exciting time getting this work done and putting it together, with the hopes of presenting this work at the upcoming Ocean Sciences meeting this February.
More to follow on this exciting final stage of my dissertation research.  ,This week I went out into the field for the very last time during my tenure at Florida State. I needed to collect sediment samples to establish my final experiment and I was joined by fellow graduate student, Rachel, and the incomparable Dr. Ingels. This was special, as Dr. Ingels supported me in the field on my first sampling effort for my dissertation, and he was there for my final field collection. As many of you are aware, record temperatures have been sweeping the United States, and this week was no different. When we got back to the car after a few hours in the field, the thermometer read 101F, and I believe it was 90F when we launched the boat. Additionally, there was no breeze during the first half of the day, which made for a pretty brutal field experience. However, a milder second-half and lots of breaks to cool down with seawater soaks and partially frozen field apparel (great idea from the field team to put some of our gear in the cooler to freeze them and cool down) made for a memorable and reasonable day on the water. In total, we collected 32 sediment cores which are now sitting in glass jars in a large aquarium at the lab awaiting further experimental conditions. These cores will remain untouched until next week, as the animals and bacteria need time to reorganize themselves (they like to live within certain layers of the mud and by transporting them to the lab, they may have been jostled around). Tomorrow I will go check on the experimental conditions and clean the aquarium. Over time, more water will evaporate from the aquarium, which will change the water's salinity (since only water evaporates there is more salt per volume of water), and I will need to carefully monitor and readjust the levels to maintain a constant environment. Additionally, I will use a gravel suction device to remove any sediment that escaped from the jars and ended up on the bottom of the aquarium tank. This happens when the jars first enter the aquarium, due to the pressure difference between the tank and the jars, and when the surface tension of the tank's water breaks, it can force small amounts of mud out of each jar. Tomorrow I will remove the excess mud from the aquarium tank, which will improve overall visibility. Next week this experiment starts in full, and I can't wait to share updates with you throughout. Stay tuned. While the seawater and microplastics are spinning away at the lab, this week I have spent much more time focusing on other aspects of my work, specifically planning and writing components. I am in the midst of working on a few research proposals for fellowship applications to fund my postdoctoral position and the proposal writing process for these positions is very different from the smaller grant applications I've written in the past. The biggest difference is that I am proposing multi-year projects but I have limited space to show the reviewers what I will be doing, the science behind the project, and the reasons why the project matters. Luckily, I have great supporters who have been helping me in this process: my prospective fellowship supervisors, mentors, peers, and of course, Dr. Ingels and my PhD committee. Right now I'm in the process of weeding through papers to extract specific information for one of these projects, as the paper that some of the ideas came from doesn't have readily available data or information. Therefore, I'm reading through how the authors did their work, finding the papers they cited, and then referring to those to piece together some background information for my own work. I have about 4 pages of writing from this process, but I think I have enough information to write the necessary pieces for my own proposal. These applications are not due until October and November, so starting early is very helpful and gives me time to complete these background research steps, get feedback on numerous drafts prior to submitting the full proposal.
Additionally, I have been writing and reviewing writing for publications. Dr. Ingels and I just published research on using nematodes as indicators of environmental health in the St. Andrew Bay system that I've been studying, and the relationship between the nematode community structures and their proximities to nearby wastewater treatment plants. The paper is accessible for free using this hyperlink. Additionally, I have reviewed another manuscript that is currently under review after its first revision. This one is based on the work I did last summer and we are hoping for good news in a few weeks. Finally, I am getting ready to head back out to the field for one last time at FSU. On Tuesday I am set to go collect my final sediment samples and I luckily roped in Dr. Ingels to come out for one more field day, as it will be our last time out on the boat together. This field collection may represent the last time that I do any sediment work, as my postdoctoral fellowships are based on larger animals that are not permanently living in or on the sediment. Stay tuned next week for hopefully one more awesome field picture of all of us covered in mud but with big bright smiles on our faces.  Today I was lucky enough to spend time in the ocean helping out with another graduate student's research. While this doesn't happen often due to the immense volume of volunteers that our elasmobranch lab has for their work, they needed an extra set of hands and I happily provided my services. The work involved setting out gill nets and pulling in the nets after 45 minutes to collect any and all animals that were caught in the nets. Today we collected multiple shark species and fishes, all for my friend's research. In this photo you can see the Atlantic sharpnose sharks we caught after they had a full workup done. My friend took measurements, blood samples, fin clips, and tissue samples to assess various aspects of the sharks' lives, and these sharks represented 84 individuals she has collected as part of this work. We were lucky enough to catch so many sharks that we finished early, as we ran out of sampling tools and supplies on the trip, though I think running out was good, as it was 100 degrees outside and I was suffering from heat exhaustion by the end of the trip. I love getting to help out my friends and fellow graduate students, as I learn new skills--like how to collect shark tissue samples--and I get to play an active role in supporting research in areas that are very different from my own work. The research activities were authorized under FWC permit SAL 1092 and supported by the FSU Coastal and Marine Laboratory. Next week will be the final week of preparation before I go out to start the sediment incubation period for my final experiment. Stay tuned to see what I'm up to and what goes into preparing a research excursion for sediment collections.  As promised, this week is all about the setup for the bioturbation and biochemical analysis experiment. Today I finished the experimental setup and built the remaining furniture pieces that I needed for this work. In the pilot experiment, I used a folding table as the aquarium stand, and quickly learned that the table was not meant to support much weight. This caused bowing in the center of the table, which also added an extra step to the photo analysis process, as I used the end of the table for the photography setup (the photographs were all on a slant and I'm not great with tripods to adjust for the slope). The new aquarium stand, pictured here, is all metal and is designed to support this aquarium. Sadly, this will be the only time I use this aquarium and stand, so I will likely sell it after the experiment is complete; our lab does not use experimental setups in the lab to answer most of our research questions. Above the aquarium tank is one of my favorite parts of the setup: the hanging arm for the air tubing. The hanging arm provides an easy way to get the airline from the cart on the left to the end of the aquarium on the right where one of the two air bubblers will reside. I added an additional side cart to hold the air pumps, temperature control, and cords, and it will serve as the photography station. I also solved some photography problems from the pilot experiment by marking each mesocosm jar at 90 degree angles to ensure that each photograph provides a consistent snapshot of each mesocosm jar. I also added a small stand to elevate the jars so that the light source can illuminate the luminophores from the below, and I marked the stand in the same way as I marked the jars so that I can line up the markings to provide even more consistency. The final, long table, is the space for the biochemistry measurements. The clip light will help provide my collaborator with some light for her work, and I will add a few more items like an air pump and manipulator stand to the table so that she has the space to do her work. Underneath the long table is a great temporary storage space for some of my items, as the temperature control system is not currently on, so the environmental chamber was close to 95 degrees today. The team at the marine lab is turning on the system so that Monday when I get to the lab the chamber will be ready for some finishing touches and for use.  This week was the last week (I think) that I will spend in the realm of deep sea research. As I have previously mentioned, I have spent some time working on side projects at the lab while I simultaneously prepare for my final experiment this summer. One of the side projects that I've featured is a continuation of research from my advisor's dissertation, and I have sorted through deep sea sediment samples to make microscope slides for identification purposes. This week, however, I finished the last of the samples for this work (to my knowledge), which also wraps up the side projects I embarked on. This timing does align with my project plans, as I will be going to collect seawater to start microplastic incubations for my final chapter. The deep sea research was exciting to help out with because shallow, coastal sediments and deep sea sediments often provide homes for animals with vastly different features. Additionally, as you'd expect, many animals in the deep sea will never be seen in coastal environments. Now, I don't know if that is the case with the animal in this week's picture, but I saw this in a sample and knew I had to take its picture, because I have no idea what it is. All I know is that it is quite large and that it has two features that remind me of a bottle cleaner brush, which I'm guessing are likely swimming appendages. As I mentioned, I am going to be spending the next few weeks working on microplastic incubations, which create microplastics that are more similar to ones in the environment, rather than sterile particles that come from the lab. Additionally, I will be testing out my new glow in the dark particles, as the most recent version of these particles did not perform as expected due to user error. I mistakenly purchased particles that were not waterproof, and most of the particles clumped and stayed at the top of the water column in my test experiments. I think I was too excited by the bright color of these particles that I didn't check on the much more important chemical features, but I should have the newest batch by tomorrow evening. Next week I will be setting up the last of the physical features for this experiment, including the aquarium stand and photography station, which will allow me to also reassess the photo quality and image analysis ease with the newly defined setup. I'm excited that this project is quickly approaching.  While I did not spend this week tracking bioturbation at the lab, I have been talking about it with groups that come to tour the lab, since bioturbation is a really important service that nematodes provide in sediments. Previously I have shown some of the pictures from the pilot but I realized that I never showed the entire microbioturbation output photographs, so that's what I'm here to do this week. Thanks to Dr. Solan in the UK, I was able to automatically extract microbioturbation data from the pilot experiment photographs, rather than manually finding each luminophore and measuring its depth. This automated process involves stitching the photographs of each mesocosm together to create a photo of all four sides of the jars. Then I use ImageJ, which is a free image analysis software, to trace the luminophore layer on the top of the sediment. This step sets a baseline for where the luminophores started and from which all measurements will be taken. Finally, I use a filtering process to isolate the luminophores in the photograph by isolating their pigments--think accentuating their color by increasing or decreasing the lighting. After this process is done, the software creates an image which has isolated the luminophores and measures the distance each particle has traveled from its starting position, assuming a vertical transport. While vertical transport isn't always the case with bioturbation, since many animals dig at angles, this method does provide the most accurate measurements of how deep particles have moved in muds, even on the finest scales. The image here is all the steps stitched together, starting with the initial image of the four sides of the mesocosm (with colors inverted in Photoshop to isolate the luminophores, shown as the red), then the filtration step which isolates luminophores as the blue, and then the generated photograph from the software with the luminophores as red dots on the black background. Additionally, this program creates an Excel spreadsheet that has all the luminophore as x,y coordinates in the final image. I can use these data to find the mean distance a luminophore has moved over the time step, the maximum distance a particle has traveled, and I can use known distance from the original image to convert these pixel measurements to centimeters, millimeters, etc. I purchased brand new luminophores to use for the full experiment and soon I will be testing these with the image analysis methods to make sure that they show up nicely. I'm hoping that these will not require the inverted color step in Photoshop, which will streamline this process even more. Thanks for attending this week's corner of arts and crafts with Aaron. Stay tuned for next week's blog.  Last week we were so busy at the lab that I didn't get a chance to post. We had two groups of students from Georgia visiting as part of our outreach opportunities for underserved communities. Alongside Dr. Ingels, Chenoah (another graduate student in the lab) and our wonderful outreach team, we took the students and their teachers into the field to study life between sediment grains and to see the larger fauna that inhabit our coastal areas and ocean. Unfortunately the weather had alternate plans for the days, but the kids had a lot of fun on the beach, in the water, and in the lab. We even caught some tardigrades with our beach sampling, and our highlight of the two days was an isopod that was incredibly fast, aptly named Flash. The tardigrade int he photo above is so hard to see because I took this picture with my phone camera, but the crystal-like structures are sand grains while the off-white shape is the tardigrade. I've also been continuing work on side projects at the lab while I pause before my research picks back up in mid-July. I am grateful for this slight break, as the weather here has been quite awful for field collections; we've had rain almost daily, with flooding and hurricane watches. However, I am ready to get back into the field to finish up this final project and I am currently working on preparing the aquarium tank for the large number of mesocosms it will house. I've decided to mark individual squares throughout the tank so that there's less bias in this work: each square and mesocosm will be numbered which will correspond to the microplastic treatment level. The treatment levels will not be labeled on the jars, which will prevent bias and/or special treatment in the data collection process.  This week I have been helping out with other projects at the lab, since I'm in between the pilot experiment and the full experiment. I will be speaking with our Swedish colleagues next week to finalize all of our plans for the full experiment, but in the meantime, I've been able to assist with two projects at the lab. The first is an assessment of California sanitation using nematodes and other meiofauna as bioindicators, and the second is an assessment of deep-sea nematodes in the UK. These projects have brought me back to my roots and it has been fun to go through deep-sea samples that are not just nematodes or recognizable meiofauna. Additionally, I've gotten to see some unexpected nematode individuals, like the one you can see in this week's photo. Notice how much larger it is than the other nematode in the picture. I definitely did not expect to see nematodes this large from the deep-sea, as nematodes tend to maximize their surface area:volume ratios. However, the size of this nematode and others from the sample not shown here may suggest that this area has plenty of organic matter within the mud that the meiofauna can feed on. Next week we have a few groups of kids coming to the marine lab to learn about meiofauna, and I will be helping them out as they explore marine sediments for the first time. I can't wait to get back into teaching and working with kids, which is always a joy, especially in times when I'm not as busy with my own research. Hopefully I can get some good pictures for you for next week's blog. See you then! No fancy title or flashy pictures for this week, because this week I finished the pilot experiment. I am very happy with how it went and I still can't believe that a few months ago I had no understanding of how aquarium systems worked or what went into maintaining tanks and seawater systems. Additionally, for anyone worried, the table survived, which is remarkable. While the results from the pilot won't go beyond the pictures I have saved on my hard drive, they set the tone for the full experiment, which will be about four times larger.
For the next month, I will be working on ways to improve the experiment, including an altered photography setup, redesigning the aquarium setup itself, and rethinking some of the sediment collection aspects. Additionally, I get to help out around the lab with additional projects and educational outreach opportunities since I have some available time. In a few weeks, I will meet with our team in Sweden to go over the results from this pilot, confirm the design and plan for the full experiment, and then get all the materials ready for my final project for my dissertation. Stay tuned for next week's update. |
 RSS Feed
RSS Feed
