|
As the title of this week's blog suggests, we faced flooding in Tallahassee this week, which prevented us from going to the lab with the students. Instead, we held virtual office hours to work with our students on finishing their final paper drafts and assisted the students with data analysis questions. While we couldn't be at the lab today, we will still go out next week to collect any final data the students are hoping to incorporate into their final papers and final oral presentations for class.
This week, I also went to the lab to clean out the remainder of my research materials. I brought back three large plastic totes (great for packing and moving) and my two research coolers that have lived at the marine lab for five years. It was quite bittersweet to clean out my final belongings and to turn the light out on my research and lab space. While I still have a few weeks left before the semester officially wraps up, I now feel like I'm truly done, with all my paperwork completed, forms signed, and belongings recovered. I've even started the process of packing and moving things out of my apartment. Where? You might be wondering. I don't know yet, but for now I am packing up and heading back to New York until the right position comes along. Next week should be a big week, with the final trip to the marine lab for my students and with the marine lab open house next weekend. I will be delaying next week's post until Saturday afternoon/evening so that I can get some pictures and updates that include the marine lab open house. You still have time to plan your schedule around the event if you are interested in attending. It's free and there will be fun for the whole family, including a scavenger hunt for the kids, food vendors, silent auction items, touch tanks, and plenty of educational materials and hands-on activities. Can't wait to see you there.
0 Comments
 This week, the students spent another week collecting data at the marine lab and began finalizing their final paper drafts. The drafts are due next week, so this week was a major push to collect as many data points as possible to maximize the strength of their statistics and interpretation. Unlike some draft papers, the final paper drafts must be completed and ready for submission, as if they couldn't edit the paper after turning it in. We will provide feedback and they will incorporate this feedback plus additional data over the next few weeks to present their finalized version of this assignment. One of the many things I love about supporting this class is that students have so much flexibility once we are at the lab and can use their time as they see appropriate. They may ask one of the TAs or faculty for writing support or questions on statistics, they might set up another run of their experiment, or they might come to the lab with the class to work on their writing out in the sunshine by the ocean. Today was a great day for writing by the ocean, as there were dolphins feeding really close to shore that were a sight to see. The three projects here represent some of the most common and the most uncommon type of project our students designed. Many students developed projects to evaluate some aspect of larval settlement, which is pictured on the left. By modifying some feature of the settlement surface (the type of material, texture, color) or the location or orientation of the settlement plates, the students are counting the number of settlers or the number of types of organisms that settle on the plates. While settlement projects are straightforward at the start and during the set up phase, they get more difficult when the students collect the data, because they are looking under the microscope for tiny larvae and moving around the entire settlement tile to find these organisms. The bottom right project is using ascidians (sea squirts) as water filters and modifying some aspect of their environment or biology to see how their filtration capacity changes. Many of the students doing this filtration-style project added phytoplankton to the water and are taking pictures of the tanks to evaluate how the shade of the green color changes over time, as a proxy for filtration capacity. Additionally, since many of the students are interested in how salinity affects filtration capacity, they are getting experience using salinity meters and fake ocean salt to create environments that are as saline as they'd like. Some have decided that perhaps their maximum salinity should be the level already at the lab and are diluting the coastal seawater to make less saline treatments. Finally, the most surprising and unique project is pictured in the top right. Two students were interested in understanding aspects of settlement and development in a manipulated setting, so they dissected the gonads from ascidians, induced spawning, fertilized the eggs, and then transplanted them to different conditions to see how development and settlement changes across a few conditions.Thank goodness the other TA studied ascidians for part of his dissertation and could help with the logistics of the project because I would not have known where to start with the students' ideas. All in all, a really productive week for the students. As for me, I've been working on editing a manuscript that I'm getting ready to submit to a journal, talking to multiple interested agencies about job prospects, and starting to prepare materials for our FSUCML Open House. Stay tuned for more updates next week.  And this time I have photos of the arts and crafts. This week was such a fun week at the lab with the students, as they continued working on their independent projects. In a few weeks, the students will present their rough drafts and then spend two weeks finishing their projects before they present their work to the class. While some of the students realized that their initial ideas were too ambitious, either because of the size of their experimental replicate or the number of samples they wanted to collect, many of the students made good progress collecting more data and some even started analyzing and visualizing their initial results. Here I'm highlighting three cool projects, and I will highlight a few more next week. On the left is a plaster cast that one student made for their project on fiddler crab burrowing. The student found a study that used plaster to measure crab burrowing activity, and they have had really great success using this method to evaluate how crab activity may changed based on the presence of visual cues. I'm highlighting this experiment because I didn't have high hopes for the plaster casting, since sediments are quite porous. I was worried that the cast wouldn't be a solid structure, but these artifacts have turned out great. In the top right is an experiment where a student is replicating features from a 1970s study on snail behaviors. These snails climb up salt marsh grasses to avoid predatory crabs, and they also climb on the grasses to feed. However, salt marsh grasses are really important to ecosystems and provide structural stability to sediments, so the degradation of the grasses can alter the landscape of the ecosystem. The student created blocks with different colored straws to evaluate if the snails are using color or shade cues to determine where to climb. I snapped this photo at the start of a trial, and the snail is currently in the center of all the straws. Finally, in the bottom right is a photograph from the early stages of a student's experiment, where the student is evaluating how salinity affects crown conch feeding activity. As global climate change affects ocean salinity, organisms may struggle to tolerate new conditions, or may experience stress. Perhaps organisms may need more energy to accommodate their stress responses. Here the student is evaluating the feeding activities of these gastropods by presenting them with shrimp (store bought, not fresh) and maintaining the conchs in different salinity environments. A reminder that if you are in the Florida big bend area, the FSU Coastal and Marine Laboratory is hosting its Open House on Saturday, April 20 from 10 am - 3 pm. All of our labs will be showcasing our research, including live animals, poster presentations, activities for kids and adults, and so much more. Come down to the lab, we'd love to see you. Tune in next week for more arts and crafts and animals updates from the students' projects.  I am happy to announce that I successfully defended my dissertation, and I was given the title of Doctor. After five years (or four and a half because this academic year isn't over) of working on my project, developing my dissertation, collecting and analyzing data, and interpreting and writing a lot of information, I have completed my journey. Although I defended this week, I still have a semester to finish [teaching] and I need to make some edits to my dissertation before I send it to the university for final approval. This week the students started their independent projects at the marine lab, and these projects will run for the next 5-6 weeks. Prior to this point, the students made observations about the organisms at the lab and their behaviors, read primary literature to learn more about what they observed, and proposed (both written and orally) an independent project to address a question raised by their research efforts. These projects concern a range of organisms, types of experiments (observational, choice/behavioral, manipulated treatments), and levels of complexity, but ultimately the students will spend the next few weeks monitoring their experiments, collecting the data, and learning how to interpret the data and present their findings. Since this course is one of the many experimental biology sections in our department, the students are expected to give multiple oral presentations and write multiple reports to meet the oral and written competency requirements. Their final written reports will be their manuscripts, as if the students were submitting their work for publication, and they will give another oral presentation discussing their findings and interpreting their results. Today, however, they needed to create settlement plates, experimental chambers, salinity treatments, and some needed to rethink their experimental design based on feedback we provided. The students learned how much arts and crafts happen behind the scenes of science experiments, as they drilled holes into materials, attached rocks to strings to weigh down their materials in the water, and created plaster casts to measure their response variables. Pictured here is a student's project in the early stages of development, as they weighed these ascidians to standardize all the treatments by the weight of the organisms to reduce confounding factors in their experiment. They will use photographic analysis of the water column in their treatment tanks to evaluate the feeding efficiency of these organisms under different conditions. Next week there will not be a blog, as it is Spring Break, so I won't have any updates, but stay tuned because I'm hoping the following week to have some great pictures from the experimental treatments that were placed into the water today. While the title is a slight overexaggeration of what I will accomplish next week, I wanted to come back after the week off from my conference to talk about the next step in my journey: my dissertation defense. This step is the last major step of a PhD program, and while it can vary widely across countries, intranational variability is minimal in this process. Here I'll lay out what the process looks like and add a few notes and resources on some differences in the process in other countries.
A dissertation defense is the last major step of a PhD program because it is the stage when the candidate presents their research to the public, responds to questions and feedback, and then has a meeting with their advisory committee to answer additional questions and feedback. The defense, therefore, has two separate aspects: the public defense or open session, and the private defense or closed session. Across many universities, these two sessions serve as a combination of two steps the candidate already accomplished: the seminar and the preliminary examinations. In the open session, a candidate presents their work in the form of a research seminar and fields questions and sometimes critiques from the audience at the end of the talk. Because this session is often open to the public, as the name implies, candidates often focus on presenting their work in a comprehensive way, and save some of the more technical information for the closed session (see below). This less jargon-filled seminar is not the approach in some European nations, where the candidate is evaluated by external adjudicators or sometimes by an opponent, who is an expert in the area of research. At the end of the open session, all attendees who are not part of the candidate's advisory committee are asked to leave so that the closed session can proceed. This session is similar to the candidate's preliminary examinations, where the committee is asking about research methods, theory, data analyses, and possibly implications of the candidate's work. Since many candidates cannot present their entire dissertation during their open session, the committee may also ask about research the candidate conducted but didn't have time to discuss with the public. Additionally, the candidate's committee usually has a copy of the dissertation well before the defense, so they have time to think about questions, pry for more information, and evaluate the candidate's thinking regarding their own research and research field. In some countries, candidates are expected to publish their full dissertation before their defense, which is an interesting concept because the publication process requires peer reviewers who critique your work and then your committee or evaluation team is critiquing you again. If you're curious about some various PhD defense formats around the world, Lantsoght (2022) summarized defense formats in 24 countries. At the end of this process, in the US at least, the candidate is ushered from the room so that the advisory committee can deliberate about the seminar and how the candidate fielded questions and critiques. At Florida State, this process also includes the committee members assigning a grade (I have no further information here), and then the candidate is told their status. Overall, this whole process lasts a few hours, and if a candidate is successful they then spend the next weeks making suggested edits to their dissertation before submitting a final version to the university. For next week's blog, expect an update, of course, and some insight into my students' first experimental day, as they will begin their independent projects. I got to hear about their methods today, and I'm really excited to see how these projects turn out in the field. Stay tuned. Lantsoght, E.O.L. (2023). Doctoral defence formats. Studies in Higher Education, 48(2), 343-355. DOI: 10.1080/03075079.2022.2137123  This week flew by, and before I knew it, it was Thursday and time for another blog. This week has featured two items: preparing for the Ocean Sciences Conference and getting my students ready for their project proposals. On Monday I learned that the department's poster printer broke, and had actually been out of commission for a while. It turns out that they don't make parts for the printer anymore, so the department is apparently working on procuring a new printer. Unfortunately, procurements take time, especially with a poster printer; these printers are designed to print high quality, glossy posters for presentations, usually 36"x48" pieces. Printing takes between 30-45 minutes, but produces a really nice finished product. As a backup, I planned to use the printing service offered by the conference (that I would have to pay for) but I found an additional poster printer for our departmental use and got my poster situation fixed by Tuesday evening. I even remembered that we had some extra poster rolls in my office, which helps protect the poster in transport and if it rains on the day I need to get the poster to the conference center. In class this week, the students started setting up pilot experiments for their projects and they started honing in on the specific question they wanted to address with their research. They will present a project proposal to us in a few weeks and then they will have a few additional weeks to start their project and collect their data. Therefore, today was a lot of problem solving tasks because while the students have really big ideas, sometimes these ideas are detrimental to the success of their projects or to the health of the animals they want to work with. In fact, one student found a published paper that used really extreme salinity levels that were most likely quite harmful to the study organisms. Luckily we talked through how they could conduct a study using similar methods but create more realistic salinity levels and less extreme upper limits. What I really enjoyed this week was asking students questions about their plans and getting to see them think through some of the potential experimental design problems they created. My hope, as is the hope of the professor and other TA, is that the students will keep their experimental design as simple as possible and conduct tidy science--science where the amount of treatment groups are limited but that there's sufficient replication and smart experimental design to account for confounding variables. I can't wait to see what the students start working on in a few weeks. Next week there will not be a blog, as I will be in the middle of the conference on Thursday and driving back to Florida on Friday. Stay tuned the week after for a special blog on what it means to defend a dissertation and what that process looks like from the candidate's perspective and from an audience member perspective. This week's picture is brought to you by our wonderful crabs at the marine lab that my students are really enjoying finding around and trying to research.  I'm really excited that I am one week closer to attending the Ocean Sciences Meeting 2024 in New Orleans with friends and colleagues from across the country. And tonight I put the finishing touches on my presentation. All presenters were asked to upload electronic versions of their posters to the community sharing page by tomorrow, and already there are so many published posters to look through. What I didn't realize was the additional accessibility that this program offers. Not only can viewers with visual impairments enlarge the text without adding pixelation, attendees can send messages directly to the poster author by clicking on a comment button, and presenters can add a tab to their poster that announces when they will have time to meet with attendees and when/where they will be presenting. These accessibility options represent a small step in the right direction for reducing and removing barriers to science communication. For me, these posters are an opportunity to view any research that I won't be able to see, either because of overlapping times or because I won't be around. While uploading an electronic version of our work is not required, I assume that many presenters have their materials ready, and the upload process is quite seamless, which should promote this additional step. For class this week the students headed back out into the field to start their independent projects. They have a few weeks before they will present a formal proposal to the class about their research question, expected results, and experimental design. This presentation will represent their second of three, and allows us (the teaching faculty and staff) to identify any concerns regarding the experimental design and methods and allows us to provide additional feedback on oral presentations. In these few weeks, we encourage the students to test out questions they are interested in, try out methods, build equipment using materials we have, and figure out what will and won't work so that they are ready to go when the independent project time officially starts. We also encourage the students to find primary literature explaining some of the observations they've made and literature that can help shape their experimental methods, as we are not experts in all the animals they want to work with. I think that many of them are excited about the freedom they get in choosing a project and potential more so with the fact that temperatures will warm up throughout the semester, so when they go to start their projects in one month, the air might be 70+ degrees. Next week I will hopefully get some good pictures of early experimental phases from the students' work, but for this week, here's a nice snapshot of the poster submission page minimized as small as possible. Even then, my computer couldn't fit more than eight posters on the screen at once. Can't wait. This week was a quiet week for research, but not for lack of work. Rather, I was exceptionally busy making edits to my dissertation, as it is due to my entire committee by Sunday. At FSU we have a 4-week policy, where PhD candidates are expected to submit their dissertation to their committee at least 4-weeks prior to the dissertation defense. This timeline allows the committee to read, critically evaluate, and prepare for the dissertation defense. I'll talk more about how a dissertation defense works in a future blog. I'm quite proud of the work I've accomplished over these past five years, and based on the initial feedback I've received from my co-advisors, I think they are as well.
Aside from preparing this dissertation, I had the joy of listening to our students give their first research presentations in class. One of my favorite (or maybe my favorite) class that we have in our biology department is the undergraduate experimental biology course that I am currently a teaching assistant for. The purpose of this course is threefold: (1) to meet the university oral communication competency requirement; (2) to meet the upper division writing requirement; and (3) to engage biology students in the practice of designing and implementing scientific experiments. To meet the oral communication requirement, our students present three times throughout the semester and receive feedback after each presentation. In this course we even give an example presentation that meets all of the rubric guidelines we use to evaluate the students. Today our students presented short talks describing a published research study, with the intention of describing the results from a figure. The next oral presentation will be the students describing their proposed research project, which is a much longer presentation, and today's talk is practice for speaking in front of a crowd and communicating scientific findings. From today's talks and the individualized feedback that we provide, our students will learn about some of their presentation habits, such as using filler words, dropping off the ends of sentences, or having non-linear presentations (presentations where ideas are forgotten and then remembered later so they are mentioned out of order). I think that this type of course should be implemented in more biology departments, because our students build important, foundational skills that make them career-ready and they get to build confidence in public speaking and writing in a welcoming environment. Over the next few weeks, we will work with the students on designing their experiments and the students will have the opportunity to test out possible methods at the marine lab. They will collect animals and plants, construct experimental chambers and treatments, and hopefully ask lots of questions as they prepare for their next presentation. I'm really excited to see what the students are interested in studying for this class, especially since two students presented on microplastics today and another on macroplastics, though both topics may be extremely difficult to study given some of the limitations for this class. I can't wait, and I hope I can provide some pictures of cool project ideas starting next week. Stay tuned!  Don't worry, though. It's not time to go just yet. This week at the lab I did finish washing and discarding all of my sediment residues, a process I convinced myself would take weeks. Nearly 800 containers of mud will now get a new home wherever the Department of Environmental Health and Safety decides is appropriate. This cleaning phase is important, as I've alluded to, because my research samples are housed in an air conditioned storage unit that is shared by all the researchers at the marine lab. These residues are therefore using space that could hold other research gear and samples so I'm hoping with their departure, other researchers can think bigger about their sampling possibilities. Importantly, though, I have only disposed of the residues. The macrofauna organisms and the remaining meiofauna still remain in conditioned storage to support ongoing and future research plans, especially regarding the environmental health and status of the St. Andrew Bay estuary, where I've conducted my dissertation. But for now, I say goodbye to our portable washing station, pictured here, that I believe could be sold or the design plans to which could be sold for a good amount of money. It's a brilliant, minimally intrusive, sink with spray nozzle attachments and extendable tables for sample washing and processing. This week for class, the students explored the different habitats we have at the marine lab. We walked in the mud, in the water, on the dry sand, and explored the docks, all the while making observations about the marine life and its behaviors. Highlights of the day included a horseshoe crab skeleton, lots of stone crabs that displayed fun behaviors, and a plethora of sea squirts, which look like slimy smaller russet potatoes grouped together. In two weeks the students will start designing their experiments and running pilot studies to test methods and the feasibility of their ideas, so today was an opportunity to see what life is like at the marine lab and what different features of the lab may be worth exploring. Finally, I officially scheduled my dissertation defense, which represents my final evaluation at Florida State, and why my parents are packing their bags to come visit. The defense is a research seminar where I will present my findings, field questions, and make the case for why my work is relevant to a broad scientific audience. My entire committee will receive a copy of my written dissertation in about a week, marking one month until my defense, and they will read through each section to both understand everything I've accomplished and give feedback and to generate points of inquiry for the defense. At my defense, my committee will have a separate session from the general audience in which to question my work, my methods, and to have me explain my thoughts regarding aspects of my work and potentially its value to the scientific community. Over the next month I will be practicing my research seminar, editing the seminar for content and ease of memorization (because I'll definitely forget things if I make it too complex), and editing my dissertation based on written feedback I receive prior to my defense. Next week will likely be a shorter update, as we are not in the field with students and I'm not sure what my own plans are, but stay tuned to find out!  This week I had the pleasure of accompanying my students out on the water for the first time. The class that I TA for is a field marine ecology course and we went out on boats this week to explore the marine life around the lab and to explore the process of marine field work. I think the students did a great job of dividing up roles, getting hands-on with the marine life, and asking questions about different organisms. The most common statement made during our trip was, "I didn't know that so many organisms were animals, especially organisms without clear mouths and eyes." Highlights of the day included seeing dolphins exploring near the boat, encountering my first sea horse in the field, and arrow crabs, which were a big hit among the students. Though close runner up was definitely the batfish, pictured here. My lab time this week involved cleaning up a few hundred of my samples. Since I have no use for the sediment residues I've amassed over the past five years, I need to start discarding them. This process involves washing out the samples from their storage containers and discarding the mud along with the fixative in which they are stored, since it is an environmental hazard. To make the process more enjoyable, though, I am stationed outside at our portable washing station, especially since I will go through all 600 or so of my samples, and the waste will fill a 40 gallon drum (or maybe multiple). This process will only take a few weeks, but it is quite a feat to look back on all the work I've done and to remind myself of all the mud I've collected and analyzed during my dissertation. I think once I'm finished I will take a picture of all the empty containers together to remind myself of all that I've accomplished and to bookmark the completion of my dissertation research. Stay tuned for that and so much more in the coming weeks. This week, like most weeks this semester, was another quiet one, since I have no additional research projects to start or finish. I couldn't make it to the lab this week because of tornado warnings in the area, so instead I focused on finishing last edits to my dissertation. That's right, this week I sent in my fully completed dissertation for first looks. While the dissertation process is not uniform, getting and incorporating feedback, just like scientists do for all our work, is integral in improving the dissertation and its readability. My co-advisors are the first to get a copy of my dissertation and once I go through a round or two of edits, I will send the updated document to the rest of my committee. Importantly, per university guidelines, the PhD committee should receive my dissertation at least 4 weeks prior to my defense. However, I'm also well aware that I am not the only student supported by many of my committee members and I don't want to put this step off longer than i need.
My next step, since I've done a lot of the preparation work, is to start synthesizing my dissertation into a defense. All PhD candidates must defend their dissertations, and while I'm not sure how the process looks in performance and creative fields, in many areas, a defense is a formal seminar with an open session and closed session. In the open session, everyone who is interested is allowed to attend, listen to the seminar, and ask questions for a set time. Then, everyone who is not on the PhD committee is asked to leave, and the PhD committee gets time with the candidate to ask more questions which may be evaluative of the entire student's journey and grasp of their work. The preparations I make for my defense will also be useful as I prepare my Ocean Sciences Meeting presentation, which will happen a few weeks earlier. This presentation will be the first time I formally present the last research chapter of my dissertation, and will be a mini practice for a subset of my dissertation defense. One major difference, though, is that my presentation at the Ocean Sciences Meeting is a poster, so rather than copying and editing slides from my defense, I will need to reformat the information and consider how I can get the attention of passerbys during the conference. Stay tuned for more updates and some pictures, as next week for the class I'm TA'ing, we are expecting to be out in the field collecting animals. Happy 2024, and I hope you had a great holiday season and a relaxing start to the new year. This week marks the last week before classes start, so I have spent my time at home writing. This will be the theme of 2024, as I have a lot of writing that I need to complete across three different themes: (1) dissertation, (2) manuscript, (3) applications.
My dissertation writing, at least for the first draft, is almost complete. I have a few final items to tidy up and then I will go through to check and edit the entire document. Without one of the chapters and its figures added, I have almost 140 pages of material. I need to add the last research chapter, but I'm waiting a few days because my collaborators and I are discussing some edits to the writing. Once I add this extra chapter, I will have a dissertation of between 150 and 165 pages, most likely, and then I get to start the larger editing process. I'm quite pleased because I've done what I think is some of the hardest aspects of the process, which is getting to this point. I have designed the experiments, conducted the research, done the reading and writing, analyzed the data, made sense of the results, and now here I am writing all my thoughts about this 5-year journey so that I can make a book out of it (yes a dissertation ends up bound as a book). I mentioned that my collaborators and I are discussing the last research chapter, as we are working on developing a manuscript to present the results. In my opinion some really cool things happened and some interesting items learned from this experiment. I wish I could say more, but for the sake of science, we will have to wait. The collaboration and discussion process will occur over the span of a few months, likely, as we want to make sure that everyone's voice is heard and that all opinions are accounted for. It is my job as the first author on the work to incorporate the feedback I find most valuable and to let authors know why I might not be incorporating their comments. Already the team has made a really interesting point about one of the trends that I didn't think about and I can't wait to explore these ideas further. Finally, I am writing a lot of job applications and postdoc applications. Today I submitted application number 17, and while many of these jobs require similar application materials, I need to customize the application to the school and talk about their curriculum, research spaces, local environment, and resources that I think I can support. This year will be full of even more applications, as there are limited microplastics research opportunities in the U.S. and it is difficult to break into the field in European countries. I am grateful for repository and/or listserv type websites that post job listings, because it is quite arduous to go to every coastal university career page to see if they have openings in your prospective department. So here's to you 2024. This year will be a whirlwind of events, and thanks to you regular readers for returning every week to see what's going on with my research. It's the end of the year, and this week at the lab was a quiet one as Dr. Ingels and I talked about how to wrap up my time at FSU. I still have another semester left, but I have finished my research and have almost 1,000 sediment residues in storage at the lab. These residues are the remnants of all the sampling I've completed and are not valuable to future research; the nematodes and other meiofauna and macrofauna I've extracted are stored separately, as researchers may be interested in conducting more work on these samples. With the year and my research wrapping up it is finally time to dispose of all the residues, which will be my task in the coming weeks. We have a large container waiting for all this mud and water, and I'd hate to be the person who has to move it when I'm finished. There are a few upsides to this major task. First, I clear up a lot of space in our storage area. While we have multiple shelves to hold samples, boxes, and materials, I am using almost two full shelves of space with just residues, which can certainly be put to better use. Additionally, I have two rolling coolers plus three to four large plastic totes holding these residues, which will make for great packing containers when I graduate.
Although my own research is finished, I will likely still help others out with their work. My roommate is one of the lead PhD candidates in the sharks and rays lab, and she always needs help out on the water with their shark surveys. I'll have more time to assist her and others with their work, but also enough piece of mind to turn down opportunities if I need a break to relax. While I won't be at the lab as frequently in the spring semester, I will continue to post weekly. These posts will likely be a mix of additional research activities, conferences updates, job and future direction updates, and highlights of cool research happening elsewhere. There will not be a post next week though, as I will be traveling and taking a mini-vacation. Stay tuned for the next blog update, coming in 2024! Have a great new year, everyone. This week was a quiet week of research, but an exciting week to plan for an exciting upcoming year. So I thought I'd take the time to highlight the cool things that are happening next year.
I was accepted to speak at Ocean Sciences Meeting 2024 in New Orleans. This conference is the flagship conference for the ocean sciences and for the broader ocean research and culture communities, and it brings together researchers from across the country to talk about latest ocean trends, new research findings, and to provide networking opportunities with government officials, universities, and non-governmental organizations. This conference is six full days of research talks, panel discussions, networking events, and community. I'm really excited for a number of talks that are offered this year, including conversations around equity and inclusion in ocean sciences and ocean science research and how to foster beneficial citizen science relationships with local communities. Additionally, some of my friends and colleagues are presenting their work, which I am excited to hear about. Interestingly, this conference has placed little emphasis on plastic pollution research, though that largely comes down to the willingness of scientists to host and plan the sessions. There are some other fun opportunities like K-12 science and undergraduate research presentations at the conference, so it will be a week of good fun. Additionally, this year I will graduate after working five years on my dissertation research. At our last meeting my PhD committee voiced no concerns with my upcoming graduation plans, which means that now I will spend time wrapping up my final data analysis, writing the research into one cohesive narrative, and evaluating all the work I completed to make sense of the past five years at the macro scale. I am also lucky that my teaching responsibility for this next semester is less time-intensive than previous ones. I will be working with students on field marine ecology during weekly trips to the marine lab as a TA for one of our many experimental biology courses. These courses fulfill an oral presentation requirement established by the university to support job-ready skills for our students. Since this teaching responsibility requires less face-to-face contact than my previous ones, I can spend the additional time writing, evaluating, and summarizing my thoughts on what the past five years have meant. Additionally, knowing how I work, I will likely take on supporting roles at the lab to help other ongoing projects. Finally, I'm excited for our bi-annual marine lab open house which will happen this April (04/20/2024). This open house is an opportunity for marine lab scientists, technicians, faculty, students, and staff to meet with the public to talk about our work, why we use the marine lab as our home base, what the Florida coastline can tell us about the world, and sometimes the open house can be an opportunity to recruit new support for the marine lab (new undergraduate assistants, lab technicians, or even graduate students). The meiolab uses this time to talk about how much we can learn from our tiny animals and how cool it is to study the small highways that exist below the sediment-water interface. If you happen to be in the area, we'd love to have you attend; it's FREE. 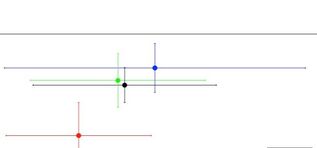 This week I started the lengthy process of data analysis and summarizing my findings from the mesocosm experiment. While data analysis can be a quick step in the relative scope of an experiment, my PhD committee impressed upon me in our recent meeting the importance of slowing down and sitting with the data to build an effective narrative. So that's what I've been doing. While non-scientists are likely familiar with the data analysis features of Microsoft Excel and potentially the add-ons you can get for your analysis needs, most scientists use Excel for organization and then import the data from the Excel spreadsheet into a more robust analysis program. One of the reasons I enjoy working outside of Excel is that Excel has limited functions regarding some of the more complex analyses I need to run, and Excel can have trouble when the data are too large. For example, the file I have for the mesocosm data is split across 17 spreadsheets, with only one of the 17 as a metadata (notes) page. Additionally, the largest spreadsheet in the file has 165 columns of data, and Excel gets slow when I need to edit some of the larger sheets. Therefore, in my work I use one of three different data programs: (1) R, (2) PRIMER, or (3) Matlab, depending on my needs (R and PRIMER for data analysis, Matlab for maps and some figures, R and PRIMER for figures). R is the very first robust data program I learned to use back in my second semester of college. I used it in my ecology laboratory class, but I was horrible with the software because we weren't taught how to code in my lecture course, while the other lecture professor incorporated it frequently into his class. So there was a large disparity across the students, which I think was apparent in the lab course. R, like some data programs, is a lot of typing out what you would like the program to do with the data and it will do it. Anything from summarizing the data and providing the mean value to running complex analyses and generating figures. Some researchers even use R for some image analysis methods they've developed. However, I have two issues with the program. First, error messages when R cannot run the code you've given it, appear quite vague. Part of that is likely that I don't understand the syntax it is using to describe the problems, but sometimes the language is quite unclear. Anytime I see an error without a clear reason, I go back through everything I typed to find out what caused the problem. This leads to the my second small complaint with R, which is that R is very much a coding program, where you have to type every item you would like R to accomplish. R does have some predictive capabilities, but it is a lot of typing, which gets frustrating if you type out lines of code and then an error happens. I do appreciate coding in R because it is quite a common data program, which means that there are great resources available on Google when I run into problems. R can also make some nice figures, like the one shown here from my mesocosm work. Notably, I have covered up a lot of the information on the figure so that my current analysis and results are not shown prior to review and publication. I use PRIMER in a situation where the data for my experiments do not follow a normal distribution even after transformation. There are assumptions we make as scientists when we analyze data, and one of the assumptions is often that the data follow a normal distribution (bell-curve). However, there are situations where this does not happen and we need to evaluate our data using an alternative method. PRIMER is great for dealing with alternative data analysis methods and it is more user friendly than other programs because it is a selective, rather than coding, program, where the user chooses what they would like the program to do to the data. PRIMER also makes nice figures, but because it uses pre-programmed click options, sometimes changes you'd like to make to figures are not possible. Finally, Matlab is a program used often by mathematicians and oceanographers, and I learned about it when I took an oceanographic modeling course during my PhD. As I'm not working on any modeling components for my current research, I don't have much use for Matlab, though it is great at making maps of sampling sites since it has detailed geographic data and local, regional, and world maps that I can alter. A lot about data this week, and while it isn't the most exciting part of my work, data analysis helps establish the narrative for my research findings and manuscripts. Next week stay tuned for an exciting post about what's happening in the new year.  Fig. 1. An unknown macrofauna individual from one of the sediment communities. It has apparent tentacles and possibly hair-like structures, and an unsegmented body. Fig. 1. An unknown macrofauna individual from one of the sediment communities. It has apparent tentacles and possibly hair-like structures, and an unsegmented body. This week at the lab I finished collecting all biomass data and identified, when possible, all macrofauna individuals from the mesocosm experiment, which means I have all the data for this project. There were some animals I could not identify, so I have pictures of each animal, and I have assigned it a unique identifier code so that any individual that is the same animal gets grouped accordingly. I will also write notes about these unidentified individuals in my data sheet so that scientists who request the raw data for the work have access to this extra information. So what's next then? I have the raw data for all the measurements from the experiment but I need to finish a few items before I can start analyzing the data. For one, I have blotted wet weight measurements for the macrofauna, and I need to convert these to dry biomass measurements. There are wonderful tools to help me in this process, so I will be using these tools to finish the biomass portion. Additionally, I need to calculate one more metric for all the data before I can evaluate the data. The bioturbation potential or community bioturbation potential, which qualifies how active fauna communities are or can be in sediments, is based on the number of animals, the types of animals, and their biomasses, hence why I need to finish the biomass data. I will also use information about the types of animals as a potential explanatory factor, because the diversity of sediment communities may be important for bioturbation or oxygen penetration. The last step, then, is to look at and analyze the data. I like this part of research because it is a story-telling process, of course based on mathematics and science, but the scientist shapes a narrative about the data and why they believe certain events happened. To tell this story, I will generate figures (graphs), tables, and statistics to help my narrative and to visualize what happened in this experiment. This process, though, can be somewhat lengthy. Especially with a lot of data and multiple variables in an experiment, it takes time to ascertain what factors lead to measured effects and then to explain why the relationships between some of the variables exist or lead to the results in the experiment. So here's to the next few weeks of working through the numbers, making figures (one of my favorite parts of science), and telling the story of what happens in sediment communities when microplastics are introduced. Stay tuned! 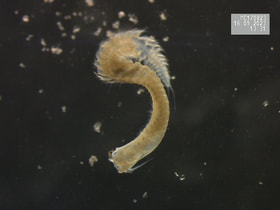 For this week, I want to spend time answer a few questions I've received recently about my research, what the role of PhD candidate is like after they graduate from their program, and what happens in research when things don't go as planned. So while this week's photo will feature a cool animal from this week's efforts, not all of these topics are related to what I accomplished in the lab. The first question I was recently asked is, "What is a postdoc, and why do you need to complete one?" A postdoc, short for postdoctoral [position], is an employment opportunity that often serves as the transition between completing a PhD and getting a faculty position at a university. Postdocs may be required in any academic field, but graduates in the natural sciences and cognitive sciences are the majority of postdoctoral scholars. While universities and hiring committees might have their own reasons for wanting applicants with postdoctoral experience, some of the common reasons include: (1) new PhDs need to gain more research experience before they start their own lab; (2) an interest in the PhD's demonstrated responsibility for large(r) sums of money; and (3) new PhDs need more first-author publications to be competitive for faculty positions. With the apparent emphasis on the value of postdoctoral experience, hopefully comes an emphasis on postdoctoral opportunities to facilitate success; unfortunately, postdoctoral funding comes from three major sources (faculty-funded opportunities, university-funded opportunities, governmental or ngo fellowship opportunities), and is a limiting factor. Finally, as postdocs are transition and growth periods, fellows are not hard committing to a specific timeline. Here I mean that even if a postdoc fellow has secured a 2-year fellowship, the individual may get hired in the first year to a faculty (or other) position, and end their fellowship early. The second question I was recently asked is, "What happens in your research when you don't know how to complete a task or the task is outside of your expertise?" For some (and maybe many) PhD students, the projects we develop and questions we want to answer are at the very edge of our comfort zones. For my own work, I had some basic marine science skills from my undergraduate work, but I had never worked on marine sediments, meiofauna, or microplastics, and I had never conducted experimental manipulations in the lab. Often our first step in tackling these research projects and tasks is reading primary literature, textbooks, and manuals, since we have documentation from so many experts before us who have tried and developed skills throughout their experience that is likely helpful in our own works. Notably, the reading doesn't stop, because we often are picking up new papers or hearing about new research that may present new ideas or alternate ways of completing our research tasks. Additionally, if possible, I try to test out the methods using samples I'm not using for my studies. The nice thing about sediment coring is that you can take practice cores where you are as long as you have the proper permits, and practicing coring skills was the first task I had when I got to FSU. Sometimes, the best method is to collaborate with the experts themselves. Besides my advisor and co-advisor, I have four great marine scientists on my PhD committee who are available to talk when I need to iron out ideas and research plans, and I can leverage connections through this team to get feedback from international researchers, especially helpful since microplastics work in the U.S. is limited. Finally, I want to close with the third question I was recently asked, which is, "What happens when things don't go as planned with research?" This happens quite a lot, whether it's a case of poor weather or boating conditions, difficulties with gear in the field, problems in the lab during or after a study, etc. I honestly think that poor weather/boating conditions and difficulties with field gear have been most of the problems I've had in my research, and both are quite frustrating. For poor weather/boating conditions, I have to reschedule with my team, which isn't ideal, but it is better to be safe than for us to try and sample under poor conditions. If we have difficulties with gear in the field, I make executive decisions on how to handle the issue and then record in my field book what happened and how we solved the problem, if necessary. A few instances our sediment core tubes have gotten stuck on the coring device, which I solved by dumping all the sediment out, using the slide weight to help remove the tube, and then I put that tube aside to never use again on the trip. This did mean dumping out a full sampling replicate and conducting an additional sampling effort, but the end result was sediment that was free from physical disturbances caused by removing the stuck tube. Problems in the lab during or after a study can be challenging because the magnitude of the problem drives the solution. If it is a situation where the power flickers and the system loses power for a brief minute (like it did in my recent work), that is a much easier situation to handle than losing the power overnight and materials thawing in a research freezer, which has happened to others. My stance on problems during or after studies is to document everything that happened with as much detail as possible, include statements regarding such anomalies in the reporting of the research, and possibly analyzing the results through two lenses--one where nothing happened or where the anomaly had no effect and one where the anomaly had an affect consistent across the study system. Thanks to everyone who sent in questions or asked questions about my work that I could feature this week. Next week I will be continuing work in the lab on measuring macrofauna biomasses and identifying the macrofauna in my sediment samples, including the polychaete pictured above. Stay tuned. 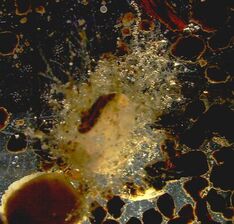 After a nice week off from research, I returned to the lab to start the macrofauna counting and biomass processes. Since we don't work with macrofauna in our lab, I was unsure how to tackle this stage of my research but I hoped that the meiofauna methods would translate to macrofauna. Thank goodness they did; I dumped all the samples into the 100-grid counting dishes we use and went through each sample to count and extract any and all macrofauna. However, since my experience with macrofauna is nearly non-existent, I decided to count and extract any organisms I immediately recognized as animals, any animal larvae, and anything I couldn't identify, but I knew wasn't sediment or plant material. While extracting extra organisms (or non-organisms) can obscure the initial macrofauna counts, I will have to readjust the counts once I have identified everything I extracted, so extra material won't be a hinderance. It is also better to do extra work now and ensure higher quality data than to ignore some unknowns and write them off as irrelevancies. 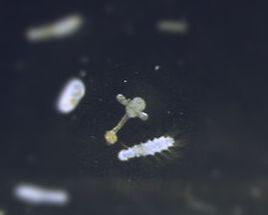 This week featured me wrapping up all of the known aspects of this final chapter of my dissertation. What I mean is that I finished counting all the meiofauna and I will finish their biomasses this weekend. After nearly 4000 individual animals, the meiofauna or known part is done. The next step is the unknown--the macrofauna. Macrofauna are the organisms that exist one size class above meiofauna. Where meiofauna are commonly defined as organisms with body sizes 32-500 micrometers, macrofauna often exist between 500 micrometers and 5 cm. While these size ranges shift depending on the environment (deep sea shifts toward smaller organisms), the rough size classification system is based on sieve mesh sizes, which is why the larger mesh size that I use in my research is 500 micrometers. This larger size retains all organisms at 500 micrometers and larger, while the meiofauna pass through the mesh and into the smaller sieve below. Once we separate the macrofauna and meiofauna by sieve size, we transfer the macrofauna to sampling jars, add a fixative, usually formaldehyde (formalin), and then store the samples away, often to never see the light of day again unless a researcher is interested in those organisms. Therefore, we do not have macrofauna expertise nor guidebooks in our lab for identifying these animals. While we can likely identify the animals to very broad taxonomic classifications (think identifying a monarch butterfly as an insect and no further), the data I need for my experimental analysis has to be more rigorous (think identifying a monarch butterfly as Genus: Danaus or as Danaus plexippus). The specificity in this chapter of my dissertation is important, as I will assign each animal a mobility and a sediment reworking score based on its life history traits. These scores have been defined by other scientists so the closer I can get to their level of specificity, the better the accuracy of my classifications. Next week I will be taking a week off from the blog as I use next week to take a short mental health break. This work has been quite tiring and I want to ensure that my work is reflective of the scientist I am, rather than reflective of my level of focus. I will spend some time identifying some of the remaining unknown meiofauna from the remaining organisms, including this little critter that looks like a fun microscopic fire hydrant. See you in two weeks.  This week in the lab I have been making microscope slides and taking more pictures to complete the biomass measurements for the meiofauna. I've resorted to calling the biomass process 'clicky clacky' as I have to click the mouse so many times to complete one measurement I'm afraid that my office mates are losing their patience. Why so many clicks? The ImageJ program has multiple methods for measuring photographs. The two most common methods are using a straight line tool and a segmented line tool, which as its name suggests, is used to measure items not in a single straight line. Since many nematodes exist in non-linear shapes, and nearly all copepods have segmented shapes, I have to click multiple times to connect line segments from their heads to tails. Additionally, ImageJ keeps track of all measurements I take in a small side window, however it assumes that each measurement is from a separate animal, so all the measurements regardless of length or width, are stored in one column. Therefore, when I import the measurements into Excel, I need to extract the width measurements, make them their own column, and then align them with the associated length measurements; again, more clicking. I am pleased to note, however, that today I finished slides for sample 24 out of 32. Between today and Tuesday--my next day at the lab--I will complete 12 samples of biomass measurements to catch up to my slide making. This means that next week will likely be my last week of slides and meiofauna biomass. I still have a few animals that I have to identify, as they are taxa I'm not familiar with, and a few copepods I have to look at under the microscope and compare their shapes with reference material (there are 8 copepod shapes which each require different conversion factors). Once this process is done, I'm one step closer to finishing this research. My interim step will be converting all of the biovolume measurements to biomass measurements using the formulas and conversions I talked about in last week's blog. Then I can move on to the macrofauna, which will be an easier biomass process, though a more difficult identification process, since we don't work with macrofauna in our lab and we don't identify them. Maybe Google reverse image search will have some ideas. |
 RSS Feed
RSS Feed
